Thermal analysis or TA refers to a variety of techniques used to measure the change in a material behavior as a function of time or temperature, either when heated, cooled, or kept at constant temperature. The sample sizes are usually in the mg range and the material changes detected are extremely small.

Examples of material behaviors that are monitored are sample weight, stiffness, melting temperature changes and sample size. These measurable changes are plotted as thermograms and the characteristics of these graphs give you precise information on fundamental material properties like melting point, glass transition and crystallization temperature. From this you can determine a material’s fundamental characteristics and composition and predict how the material will behave in each application.
Using thermal analysis is important to make sure materials meet specifications. It’s commonly used in research and development, and quality control in many industries including polymer, pharmaceutical, food, electronics, battery materials, ceramics, and metals.
A thermal analysis instrument consists of a furnace and a sample holder which includes a sensor. As you either heat or cool the sample in the furnace, the sensor detects the changes in the sample’s thermal or physical properties. The temperature control unit controls the furnace temperature whilst the data recording unit records the signals of the sensors and sample temperature and analyses them.
Different type of furnaces and cooling accessories are available depending on the required temperature range. These measurements are typically done in a control atmosphere using a purged gas typically consisting of nitrogen or air.
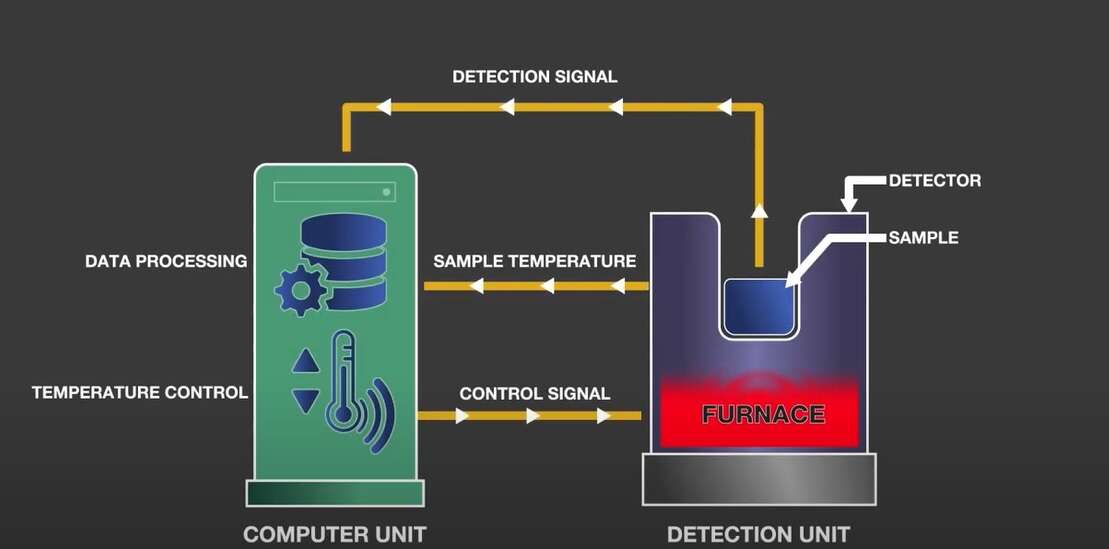
The temperature control, data recording and analysis are all computer-controlled. Depending on the information required, the user will select one or multiple thermal analysis techniques. The computer can be connected to several instruments which have the other types of measurement techniques, enabling simultaneous measurement and analysis.
There are different measuring methods available, and the right method depends on which thermal properties are being analyzed. The most common methods are: DSC – differential scanning calorimetry, TGA – thermogravimetric analysis, DMA – dynamic mechanical analysis and TMA – thermomechanical analysis.
Each thermal analyzer can tell you a great deal about a material’s properties but sometimes the results and evaluation can be ambiguous from one technique to another. Depending on the information required, one or multiple techniques will need to be selected.
| Melting | ★★★ | ★★☆ | ★☆☆ | ★☆☆ |
| Glass Transition | ★★★ | ★☆☆ | ★★★ | ★★★ |
| Sample visualization | ★★★ | ★★★ | N/A | ★★★ |
| Crystalization | ★★★ | ★☆☆ | ★★☆ | ★★☆ |
| Reaction (curing polymerization) | ★★★ | ★★☆ | ★★★ | ★★★ |
| Sublimation, evaporation, dehydration | ★☆☆ | ★★★ | N/A | ★☆☆ |
| Thermal absorbtion / adsorbtion | N/A | ★★★ | ★☆☆ | ★☆☆ |
| Thermal decomposition | ★☆☆ | ★★★ | N/A | N/A |
| Thermal expansion and contraction | ★★☆ | ★★☆ | ★★★ | ★★★ |
| Study of thermal history | ★★★ | ★★☆ | ★★★ | ★★★ |
| Cp (specific heat capacity) | ★★★ | ★★★ | N/A | N/A |
| Dynamic viscoelasticity | N/A | N/A | ★★☆ | ★★★ |
| Evolved gas analysis | N/A | ★★★ | N/A | N/A |
| Frequency dependent event | N/A | N/A | ★☆☆ | ★★★ |
| Kinetic study | ★★★ | ★★★ | N/A | N/A |
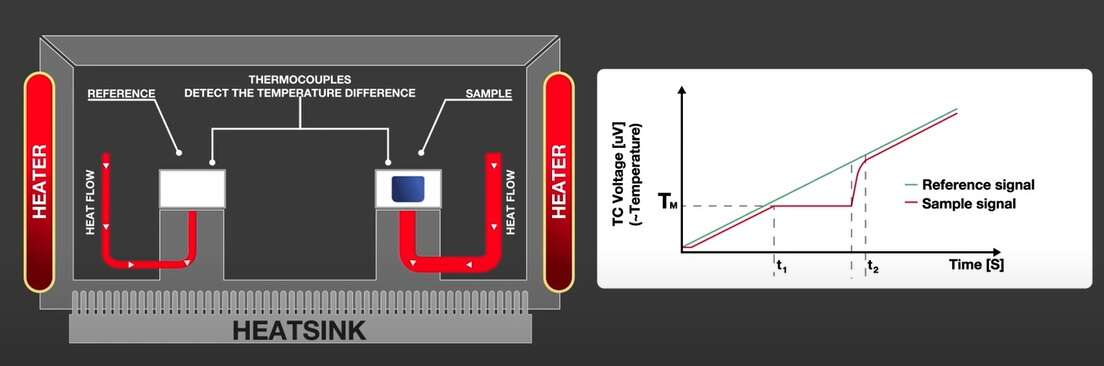
DSC measures heat flow required to go through a thermal event. These endothermic or exothermic events give information about thermal properties such as melting, glass transition, crystallization, chemical reactions, thermal history, and to determine specific heat capacity.
The sample to be analyzed and a reference (typically an empty pan) are placed in the DSC furnace and the temperature of the furnace is changed under controlled conditions. The temperature difference between the sample and the reference material is measured as a function of the applied temperature.
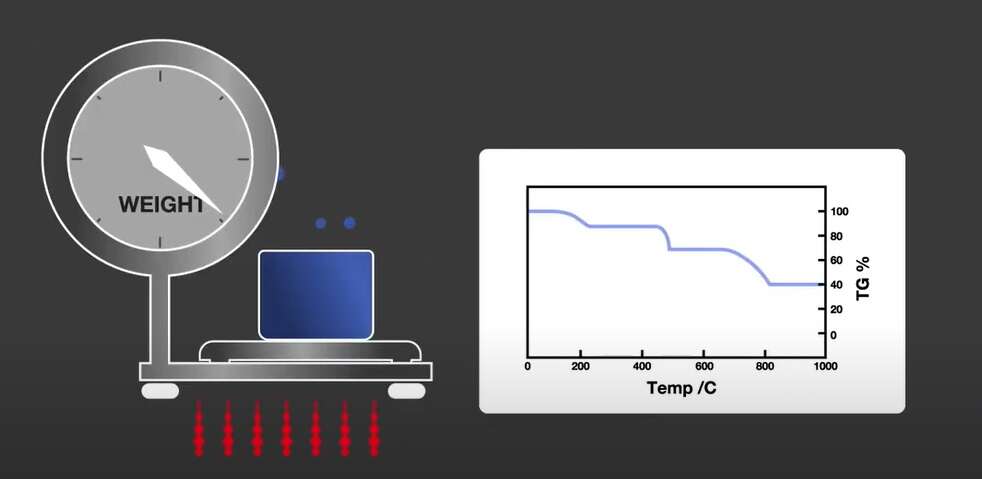
TGA measures sample mass change over time or temperature. It’s used for evaluating thermal stability, decomposition temperature and quantitative analysis of components. Some TGA can combine a DSC and TGA into one simultaneous single unit, to give you a STA, a simultaneous thermogravimetric analyzer.
The sample is placed in a thermogravimetric furnace on a highly sensitive balance. The mass of the sample is monitored against time or controlled temperature in standard or an inert atmosphere.
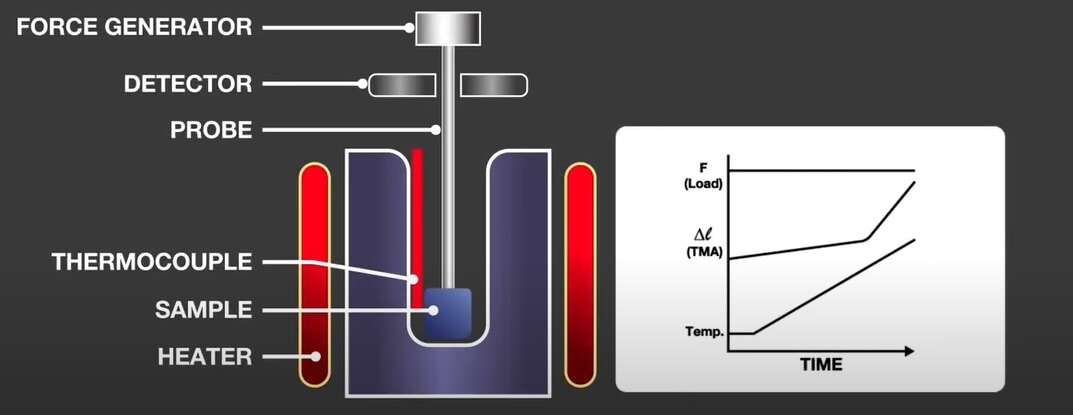
TMA measures change in sample dimensions as a function of time or temperature. Phenomena caused by deformation includes thermal expansion, shrinkage, glass transition, and the curing reaction can be measured with this technique.
A sample is placed in a TMA furnace where it’s subject to a force (compression, tension, flexure, or torsion) while the temperature of the sample is changed in a controlled way. Any deformation of the sample at a specific temperature is detected.
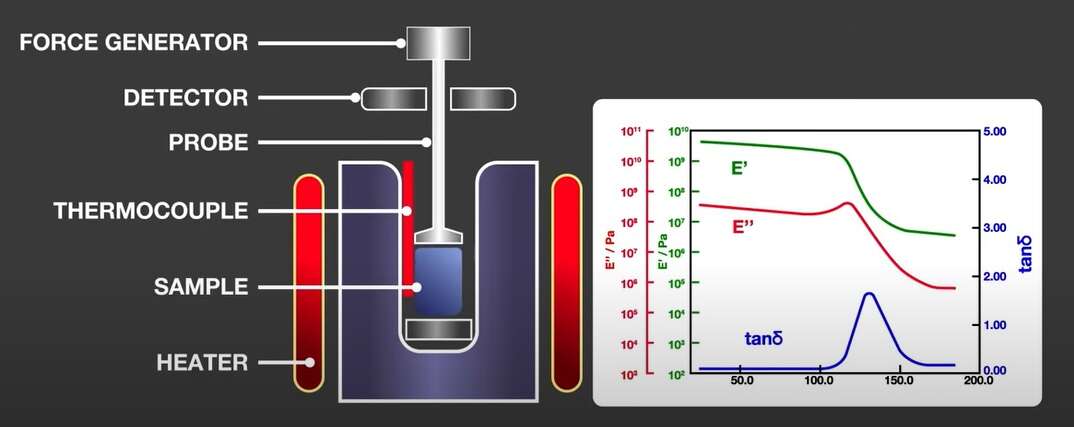
DMA measures the viscoelastic properties of materials. Its main use is for glass transition detection, but it can also be used to measure secondary transition, materials stiffness, curing level, and damping.
A sample is clamped in the measurement head of the DMA instrument and a sinusoidal force is applied while the temperature of the sample is changed in a controlled way. The relation between the applied force and the resulting deformation is measured.
The main advantage of thermal analysis is that it precisely analyses fundamental material properties. Even for complex materials you can often tease out the behavior of the constituent polymers to ascertain what’s in the mix. It’s applicable to a wide range of materials and doesn’t require material specific calibration curves, so you can easily investigate novel materials.
There is very little sample preparation, with no harmful chemicals to contend with and, with a little training, the analyses can be run by anyone, especially with an instrument that has a high degree of automation.
The analysis procedures are relatively short, with many completed in under an hour. The equipment is inexpensive to run, and you don’t have to keep the equipment in standby when not in use, cutting electricity and gas consumption.
Ultimately, thermal analyzers easy to use and plenty of accredited methods are available to make sure you get relevant information for your products.
How can we help you?
We offer the full range of thermal analyzers, DSC, STA, TMA and DMA. Our experts are more than happy to answer any questions you may have about thermal analysis. Please get in touch with us.
Contact us
You might also be interested: